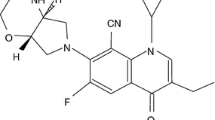
Abstract
-
▴ Moxifloxacin is a new fluoroquinolone antibacterial agent with a broad spectrum of activity, encompassing Gram-negative and Gram-positive bacteria. It has improved activity against Gram-positive species (including staphylococci, streptococci, enterococci) and anaerobes compared with ciprofloxacin. This is offset by slightly lower activity against pseudomonal species and Enterobacteriaceae.
-
▴ In common with other fluoroquinolones, moxifloxacin attains good penetration into respiratory tissues and fluids and its bioavailability is substantially reduced by coadministration with an antacid or iron preparation. However, moxifloxacin does not interact with theophylline or warfarin.
-
▴ In clinical trials in patients with community-acquired pneumococcal pneumonia (CAP), acute exacerbations of chronic bronchitis (AECB) or acute sinusitis, moxifloxacin 400mg once daily achieved bacteriological and/or clinical success rates of approximately 90% or higher.
-
▴ Moxifloxacin was as effective as amoxicillin 1g 3 times daily and clarithromycin 500mg twice daily in CAP and as effective as clarithromycin in AECB. In patients with sinusitis, a 7-day course of moxifloxacin 400mg once daily was as effective as a 10-day course of cefuroxime axetil 250mg twice daily.
-
▴ In contrast to some other fluoroquinolones, moxifloxacin appears to have a low propensity for causing phototoxic and CNS excitatory effects. The most common adverse events are gastrointestinal disturbances.
Similar content being viewed by others
References
Bauernfeind A. Comparison of the antibacterial activities of the quinolones Bay 12-8039, gatifloxacin (AM 1155), trovafloxacin, clinafloxacin, levofloxacin and ciprofloxacin. J Antimicrob Chemother 1997; 40: 639–51
Krasemann C, Meyer JM, Springsklee M. Suggested breakpoints for moxifloxacin (MXF). Poster (#PO206) accepted by 9th ECCMID; 1999 Mar 21–24; Berlin
Fass RJ. In. vitro activity of Bay 12-8039, a new 8-methoxyquinolone. Antimicrob Agents Chemother 1997 Aug; 41: 1818–24
Schülin T, Wennersten CB, Ferraro MJ, et al. Susceptibilities of Legionella spp. to newer antimicrobials in vitro. Antimicrob Agents Chemother 1998 Jun; 42: 1520–3
Bébéar CM, Renaudin H, Boudjadja A, et al. In vitro activity of BAY 12-8039, a new fluoroquinolone, against mycoplasmas. Antimicrob Agents Chemother 1998 Mar; 42: 703–4
Ruckdeschel G, Lob S. In vitro activity of a new 8-methoxyquinolone, BAY 12-8039, against Legionella spp. in comparison to ciprofloxacin, erythromycin and rifampicin [abstract]. 36th ICAAC; 1996 Sep 15; New Orleans, 101
Roblin PM, Hammerschlag MR. In vitro activity of a new 8-methoxyquinolone, BAY 12-8039, against Chlamydia pneumoniae. Antimicrob Agents Chemother 1998 Apr; 42: 951–2
Biedenbach DJ, Barrett MS, Croco MAT, et al. BAY 12-8039, a novel fluoroquinolone: activity against important respiratory tract pathogens. Diagn Microbiol Infect Dis 1998 Sep; 32: 45–50
Weiss K, Laverdiere M, Restieri C. Comparative activity of trovafloxacin and Bay 12-8039 against 452 clinical isolates of Streptococcus pneumoniae. J Antimicrob Chemother 1998 Oct; 42: 523–5
Klugman KP, Capper T. Concentration-dependent killing of antibiotic-resistant pneumococci by the methoxyquinolone moxifloxacin. J Antimicrob Chemother 1997 Dec; 40: 797–802
Ji B, Lounis N, Maslo C, et al. In vitro and in vivo activities of moxifloxacin and clinafloxacin against Mycobacterium tuberculosis. Antimicrob Agents Chemother 1998 Aug; 42: 2066–9
Aldridge KE, Ashcraft DS. Comparison of the in vitro activities of Bay 12-8039, a new quinolone, and other antimicrobials against clinically important anaerobes. Antimicrob Agents Chemother 1997 Mar; 41: 709–11
Edlund C, Sabouri S, Nord CE. Comparative in vitro activity of BAY 12-8039 and five other antimicrobial agents against anaerobic bacteria. Eur J Clin Microbiol Infect Dis 1998 Mar; 17: 193–5
Woodcock JM, Andrews JM, Boswell FJ, et al. In vitro activity of BAY 12-8039, a new fluoroquinolone. Antimicrob Agents Chemother 1997 Jan; 41: 101–6
Boswell FJ, Andrews JM, Wise R. Pharmacodynamic properties of BAY 12-8039 on Gram-positive and Gram-negative organisms as demonstrated by studies of time-kill kinetics and post-antibiotic effect. Antimicrob Agents Chemother 1997 Jun; 41: 1377–9
Al-Nawas B, Shah PM. Intracellular activity of ciprofloxacin and moxifloxacin, a new 8-methoxyquinolone, against methicillin-resistant Staphylococcus aureus. J Antimicrob Chemother 1998 Jun; 41: 655–8
Davidson RJ, Fuller J, Low DE. Pharmacodynamic properties of moxifloxacin, levofloxacin, and clarithromycin in S. pneumoniae and H. influenzae [abstract]. 38th ICAAC; Sep 24; San Diego; 227
Jacobs E, Dalhoff A, Brunner H. Efficacy of BAY 12-8039 in Mycoplasma pneumoniae infected guinea pigs [abstract]. 36th ICAAC; 1996 Sep 15; New Orleans, 102
Rouse MS, Piper KE, Patel R, et al. In vitro and in vivo activity of BAY12-8039 or trovafloxacin against penicillin-resistant Streptococcus pneumoniae experimental pneumonia in immunocompetent mice [abstract]. 36th ICAAC 1996 Sep 15; New Orleans: 29
Waterbury K, Wang JJ, Barbiero M, et al. Efficacy of BAY 12-8039, a potent new quinolone, in mouse models of typical and atypical respiratory infection [abstract]. 36th ICAAC 1996 Sep 15; New Orleans: 103
Miyazaki E, Chaisson RE, Bishai WR. In vivo activity of B AY12-8039, a new 8-methoxy-quinolone, in a mouse model of tuberculosis [abstract]. 38th ICAAC 1998; Sep 24; San Diego: 230
Schmidt H, Dalhoff A, Stuertz K, et al. Moxifloxacin in the therapy of experimental pneumococcal meningitis. Antimicrob Agents Chemother 1998 Jun; 42: 1397–401
Ostergaard C, Sorensen TK, Knudsen JD, et al. Evaluation of moxifloxacin, a new 8-methoxyquinolone, for treatment of meningitis caused by a penicillin-resistant Pneumococcus in rabbits. Antimicrob Agents Chemother 1998 Jul; 42: 1706–12
Dalhoff A. Dissociated resistance among quinolones. Poster presented at ICIDs 1998; May 15–18; Boston
Souli M, Wennersten CB, Eliopoulos GM. In vitro activity of BAY 12-8039, a new fluoroquinolone against species representative of respiratory tract pathogens. Int J Antimicrob Agents 1998; 10(1): 23–30
Tankovic J, Bechoual R, Leclercq L, et al. In vitro activity of BAY 12-8039 against fluoroquinolone-resistant Gram negative bacilli and Enterococcus faecalis. Poster presented at the 37th ICAAC; 1997 Sep 28; Toronto, 169
Schmitz F-J, Hofmann B, Hansen B, et al. Relationship between ciprofloxacin, ofloxacin, levofloxacin, sparfloxacin and moxifloxacin (BAY 12-8039) MICs and mutations in gr1A, gr1B, gyrA and gyrB in 116 unrelated clinical isolates of Staphylococcus aureus. J Antimicrob Chemother 1998 Apr; 41: 481–4
Dalhoff A, Heidtmann M, Obertegger S, et al. Lack of in vivo emergence of resistance against BAY 12-08039 in Staph. aureus and Strep pneumoniae [abstract]. 8th ICIDs 1998; May 15: 124
Stass H, Dalhoff A, Kubitza D, et al. Pharmacokinetics, safety, and tolerability of ascending single doses of moxifloxacin, a new 8-methoxy quinolone, administered to healthy subjects. Antimicrob Agents Chemother 1998 Aug; 42: 2060–5
Sullivan JT, Woodruff M, Lettieri J, et al. Pharmacokinetics (PK) and tolerability of the new methoxyquinolone BAY 12-8039: 10 days’ treatment at 400 mg daily. Poster presented at the 8th ECCMID; 1997 25 May; Lausanne, 4
Stass HH, Kubitza D. Cross over study to assess absolute bioavailability (AB) and absorption characteristics (AC) of moxifloxacin in man. Poster presented at the 8th ICIDs; 1998 May 15–18; Boston
Stass H, Brunner M, Eichler HG, et al. Comparison of skin blister fluid (SBF) and interstitial tissue kinetics of BAY 12-8039 in healthy volunteers (HV) using microdialysis (Mi) [abstract]. 38th ICAAC; 1998 Sep 24; San Diego, 4
Stass H, Kubitza D. Study to assess the interaction between moxifloxacin (M) and dairy products (DP) in healthy volunteers (HV). Poster presented at the 2nd European Congress of Chemotherapy and 7th Biennial Conference of Antiinefective Agents and Chemotherapy, May 10–13, 1998, Hamburg
Andrews J, Honeybourne D, Jevons G, et al. Penetration of Bay 12-8039 into bronchial mucosa, epithelial lining [abstract]. 38th ICAAC; 1998 Sep, San Diego 24, 9
Gehanno P, Stass H, Arvis P. Penetration of moxifloxacin (MFX) into sinus tissues following multiple oral dosing. Abstract accepted by 9th ECCMID; 1999 Mar 21–24; Berlin
Siefert HM, Daehler HP, Goeller G, et al. BAY 12-8039, a new 8-methoxyquinolone: investigations on distribution in rats [abstract]. 37th ICAA; 1997 Sep 28; Toronto, 171
Siefert HM, Bühner K, Domdey-Bette A, et al. BAY 12-8039, a new 8-methoxy-quinolone: pharmacokinetics in rats and monkeys [abstract]. 36th ICAAC 1996 Sep 15; New Orleans: 103
Data on file, Bayer.
Kern A, Kanhai W, Fröhde R, et al. BAY 12-8039, a new 8-methoxy-quinolone: metabolism in rat, monkey, and man [abstract]. 36th ICAAC; 1996 Sep 15; New Orleans, 103
Kubitza D, Stass HH, Wingender W, et al. BAY 12-8039 (I), a new 8-methoxy-quinolone: safety (S), tolerability and steady state pharmacokinetics (PK) in healthy male volunteers [abstract]. 36th ICAAC; 1996 Sep 15; New Orleans, 104
Sullivan JT, Lettieri J, Hogan CH, et al. Age and gender effects on pharmacokinetics (PK) of BAY 12-8039 [abstract]. 98th American Society for Clinical Pharmacology and Therapeutics; 1997 Mar 5; San Diego, 148
Stass H, Halabi A, Delesen H. No dose adjustment needed for patients with renal impairment receiving oral BAY 12-8039 (M) [abstract]. 38th ICAAC; 1998 Sep 24; San Diego, 5
Data on file, Bayer.
Petipretz P, Branco Pires J, Dosedel J, et al. Moxifloxacin (MFX) versus amoxicillin (AMOX) in the treatment of community-acquired suspected pneumococcal pneumonia: a multinational double-blind randomised study. Abstract accepted by 9th ECCMID; 1999 Mar 21–24; Berlin
Krasemann C, Meyer JM, Springkslee M. Moxifloxacin (MFX) in community-acquired pneumonia - a bacteriological and clinical meta-analysis. Poster (#PO205) accepted by 9th ECCMID; 1999 Mar 21–24, Berlin
Wilson R, Kubin R. Short-course moxifloxacin (MFX) vs clarithromycin (CLAR) in acute exacerbations of chronic bronchitis (AECB). Poster (#PO209) accepted by 9th ECCMID; 1999 Mar 21–24; Berlin
Krasemann C, Meyer JM, Springsklee M. Moxifloxacin (MFX) in acute exacerbations of chronic bronchitis - a bacteriological and clinical meta-analysis. Poster (#PO203) accepted by 9th ECCMID; 1999 Mar 21–24; Berlin
Siegert R, Gehanno P, Nikolaidis P, et al. A comparison of the safety and efficacy of moxifloxacin (BAY 12-8039) and cefuroxime axetil in the treatment of acute bacterial sinusitis in adults. Bayer, data on file
Krasemann C, Meyer JM, Springsklee M. Moxifloxacin in acute sinusitis - a bacteriological and clinical meta-analysis. Poster (#PO204) accepted by 9th ECCMID; 1999 Mar 21–29; Berlin
Springsklee M, Reiter C, Meyer JM. Safety and tolerability profile of MFX. Poster (#PO208) accepted by 9th ECCMID; 1999 Mar 21–29; Berlin
Sullivan JT, Kubitza D, Schuhly U, et al. Safety of the new quinolone BAY 12-8039 in 130 healthy volunteers [abstract]. 20th ICC; 1997 Jun 29; Sydney, 108
Schmuck G, Schurmann A, Schluter G. Determination of the excitatory potency of different fluoroquinolones in the central nervous system by an in vitro model. Antimicrobial Agents and Chemotherapy 1997; 42: 1831–6
Ferguson J, Al-Ajmi H, Kubin R, et al. A double-blind, placebo and lomefloxacin controlled human volunteer phototest study to determine the photosensitising potential of oral moxifloxacin (BAY 12-8039) [abstract]. 8th ICID 1998; 15 May, Boston, 197
Vohr H–W, Wasinska-Kempka G, Ahr HJ. Studies on the phototoxic potential of a new 8-methoxy-quinolone: BAY 12-8039 [abstract]. 36th ICAAC 1996 Sep 15; New Orleans: 103
Stass HH, Boettcher M, Horstmann R. Study to evaluate the interaction between BAY 12-8039 (BA) and antacids (AN) [abstract no. 3358]. 20th ICC; 1997 Jun 29–Jul 3, Sydney, 109
Stass H, Kubitza D. Study to evaluate the interaction between moxifloxacin (M) and iron supplements (FE) [abstract no. T154]. 2nd European Congress of Chemotherapy and 7th Biennial Conference on Antiinfective Agents and Chemotherapy; 1998 May 10-13, Hamburg
Stass HH, Kubitza D, Schwietert R, et al. BAY 12-8039 (BA) does not interact with theophylline (TH) [abstract no. 3356]. 20th ICC, Jun 29–Jul 3, 1997, Sydney: 108
Stass HH, Dietrich H, Sachse R. Influence of a four-times dosing of 500 mg probenecid on kinetics of BAY 12-8039 after administration of a single 400 mg dose in healthy male volunteers [abstract no. F-154]. 37th ICAAC; 1997 28 Sep, Toronto: 172
Stass HH, Ochmann K. Study to evaluate the interaction between BAY 12-8039 (BA) and ranitidine (RA) [abstract no. 3357]. 20th ICC; 1997 Jun 29–Jul 3; Sydney, 108
Müller FO, Hundt HKL, Muir AR, et al. Study to investigate the influence of 400 mg BAY 12-8039 given once daily to healthy volunteers on PK and PD of warfarin (W) [abstract]. 38th ICAAC; 1998 Sep 24; San Diego, 4
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balfour, J.A.B., Wiseman, L.R. Moxifloxacin. Drugs 57, 363–373 (1999). https://doi.org/10.2165/00003495-199957030-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199957030-00007