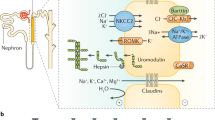
Abstract
Cardiovascular problems are a major cause of morbidity and mortality in patients with autosomal-dominant polycystic kidney disease (ADPKD). Hypertension is a common early symptom of ADPKD, and occurs in approximately 60% of patients before renal function has become impaired. Hypertension is associated with an increased rate of progression to end-stage renal disease and is the most important potentially treatable variable in ADPKD. Left ventricular hypertrophy, which is a powerful, independent risk factor for cardiovascular morbidity and mortality, also occurs frequently in patients with ADPKD. Both hypertension and left ventricular hypertrophy have important roles in cardiovascular complications in these individuals. Moreover, biventricular diastolic dysfunction, endothelial dysfunction, increased carotid intima–media thickness, and impaired coronary flow velocity reserve are present even in young patients with ADPKD who have normal blood pressure and well-preserved renal function. These findings suggest that cardiovascular involvement starts very early in the course of ADPKD. Intracranial and extracranial aneurysms and cardiac valvular defects are other potential cardiovascular problems in patients with ADPKD. Early diagnosis and treatment of hypertension, with drugs that block the renin–angiotensin–aldosterone system, has the potential to decrease the cardiovascular complications and slow the progression of renal disease in ADPKD.
Key Points
-
Cardiovascular problems are a major cause of morbidity and mortality in patients with autosomal-dominant polycystic kidney disease (ADPKD)
-
Hypertension, a common symptom of ADPKD, is associated with rapid progression to end-stage renal disease; the renin–angiotensin–aldosterone system (RAAS) is important in the development of hypertension in this setting
-
Early vascular changes have been reported even in young patients with ADPKD and normal blood pressure
-
Left ventricular hypertrophy is a common finding in patients with ADPKD
-
Patients with ADPKD have a higher prevalence of aneurysms and cardiac valvular abnormalities than the general population
-
Early and effective treatment of hypertension is very important to decrease the morbidity and mortality of patients with ADPKD, and drugs that inhibit the RAAS might be beneficial in this context
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ecder, T. et al. in Diseases of the Kidney and Urinary Tract (ed. Schrier, R. W.) 502–539. (Lippincott Williams & Wilkins, Philadelphia, 2007).
PKD Foundation. Polycystic Kidney Disease: The Most Common Life-Threatening Genetic Disease. (Polycystic Kidney Research Foundation, Kansas City, 2000).
Fick, G. M. et al. Causes of death in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 5, 2048–2056 (1995).
Perrone, R. D. et al. Survival after end-stage renal disease in autosomal dominant polycystic kidney disease: contribution of extrarenal complications to mortality. Am. J. Kidney Dis. 38, 777–784 (2001).
Chapman, A. B. & Schrier, R. W. Pathogenesis of hypertension in autosomal dominant polycystic kidney disease. Semin. Nephrol. 11, 653–660 (1991).
Ecder, T. & Schrier, R. W. Hypertension in autosomal-dominant polycystic kidney disease: early occurrence and unique aspects. J. Am. Soc. Nephrol. 12, 194–200 (2001).
Gabow, P. A. et al. Factors affecting the progression of renal disease in autosomal-dominant polycystic kidney disease. Kidney Int. 41, 1311–1319 (1992).
Chapman, A. B. et al. Left ventricular hypertrophy in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 8, 1292–1297 (1997).
Kelleher, C. L. et al. Characteristics of hypertension in young adults with autosomal dominant polycystic kidney disease compared with the general U.S. population. Am. J. Hypertens. 17, 1029–1034 (2004).
Schrier, R. W. et al. The role of parental hypertension in the frequency and age of diagnosis of hypertension in offspring with autosomal-dominant polycystic kidney disease. Kidney Int. 64, 1792–1799 (2003).
Sedman, A. et al. Autosomal dominant polycystic kidney disease in childhood: a longitudinal study. Kidney Int. 31, 1000–1005 (1987).
Fick, G. M. et al. The spectrum of autosomal dominant polycystic kidney disease in children. J. Am. Soc. Nephrol. 4, 1654–1660 (1994).
Ivy, D. D. et al. Cardiovascular abnormalities in children with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 5, 2032–2036 (1995).
Shamshirsaz, A. et al. Autosomal-dominant polycystic kidney disease in infancy and childhood: progression and outcome. Kidney Int. 68, 2218–2224 (2005).
Gabow, P. A. et al. Renal structure and hypertension in autosomal dominant polycystic kidney disease. Kidney Int. 38, 1177–1180 (1990).
Graham, P. C. & Lindop, G. B. M. The anatomy of the renin-secreting cell in adult polycystic kidney disease. Kidney Int. 33, 1084–1090 (1988).
Torres, V. E. et al. Synthesis of renin by tubulocystic epithelium in autosomal-dominant polycystic kidney disease. Kidney Int. 42, 364–373 (1992).
Loghman-Adham, M. et al. The intrarenal renin-angiotensin system in autosomal dominant polycystic kidney disease. Am. J. Physiol. Renal Physiol. 287, F775–F788 (2004).
Chapman, A. B. et al. The renin–angiotensin–aldosterone system and autosomal dominant polycystic kidney disease. N. Engl. J. Med. 323, 1091–1096 (1990).
Torres, V. E. et al. Effect of inhibition of converting enzyme on renal hemodynamics and sodium management in polycystic kidney disease. Mayo Clin. Proc. 66, 1010–1017 (1991).
Watson, M. L. et al. Effects of angiotensin converting enzyme inhibition in adult polycystic kidney disease. Kidney Int. 41, 206–210 (1991).
Doulton, T. W. et al. The effect of sodium and angiotensin-converting enzyme inhibition on the classic circulating renin–angiotensin system in autosomal-dominant polycystic kidney disease patients. J. Hypertens. 24, 939–945 (2006).
Harrap, S. B. et al. Renal, cardiovascular and hormonal characteristics of young adults with autosomal dominant polycystic kidney disease. Kidney Int. 40, 501–508 (1991).
Barrett, B. J. et al. Differences in hormonal and renal vascular responses between normotensive patients with autosomal dominant polycystic kidney disease and unaffected family members. Kidney Int. 46, 1118–1123 (1994).
Torres, V. E. et al. Natriuretic response to volume expansion in polycystic kidney disease. Mayo Clin. Proc. 64, 509–515 (1989).
Klein, I. H. et al. Sympathetic activity is increased in polycystic kidney disease and is associated with hypertension. J. Am. Soc. Nephrol. 12, 2427–2433 (2001).
Bakris, G. et al. Role of vasopressin in essential hypertension: racial differences. J. Hypertens. 15, 545–550 (1997).
Fernandes, S. et al. Chronic V2 vasopressin receptor stimulation increases basal blood pressure and exacerbates deoxycorticosterone acetate-salt hypertension. Endocrinology 143, 2759–2766 (2002).
Torres, V. E. Vasopressin antagonists in polycystic kidney disease. Kidney Int. 68, 2405–2418 (2005).
Danielsen, H. et al. Expansion of extracellular volume in early polycystic kidney disease. Acta Med. Scand. 219, 399–405 (1986).
Hocher, B. et al. Renal endothelin system in polycystic kidney disease. J. Am. Soc. Nephrol. 9, 1169–1177 (1998).
Munemura, C. et al. Epidermal growth factor and endothelin in cyst fluid from autosomal dominant polycystic kidney disease cases: Possible evidence of heterogeneity in cystogenesis. Am. J. Kidney Dis. 24, 561–568 (1994).
Giusti, R. et al. Plasma concentration of endothelin and arterial pressure in patients with ADPKD. Contrib. Nephrol. 115, 118–121 (1995).
Wang, D. et al. Endothelium-dependent relaxation of small resistance vessels is impaired in patients with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 11, 1371–1376 (2000).
Wang, D. et al. Endothelial dysfunction and reduced nitric oxide in resistance arteries in autosomal dominant polycystic kidney disease. Kidney Int. 64, 1381–1388 (2003).
Al-Nimri, M. A. et al. Endothelium-derived vasoactive mediators in polycystic kidney disease. Kidney Int. 63, 1776–1784 (2003).
Merta, M. et al. Role of endothelin and nitric oxide in the pathogenesis of arterial hypertension in autosomal dominant polycystic kidney disease. Physiol. Res. 52, 433–437 (2003).
Wang, D. et al. Asymmetric dimethylarginine and lipid peroxidation products in early autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 51, 184–191 (2008).
Kocaman, O. et al. Endothelial dysfunction and increased carotid intima–media thickness in patients with autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 43, 854–860 (2004).
Turkmen, K. et al. Coronary flow velocity reserve and carotid intima media thickness in patients with autosomal dominant polycystic kidney disease: from impaired tubules to impaired carotid and coronary arteries. Clin. J. Am. Soc. Nephrol. 3, 986–991 (2008).
Borresen, M. L. et al. Pulse wave reflection is amplified in normotensive patients with autosomal-dominant polycystic kidney disease and normal renal function. Am. J. Nephrol. 27, 240–246 (2007).
Griffin, M. D. et al. Vascular expression of polycystin. J. Am. Soc. Nephrol. 8, 616–626 (1997).
Torres, V. E. et al. Vascular expression of polycystin-2. J. Am. Soc. Nephrol. 12, 1–9 (2001).
Koren, M. J. et al. Relationship of left ventricular mass and geometry to morbidity and mortality in uncomplicated essential hypertension. Ann. Intern. Med. 114, 345–352 (1991).
Zeier, M. et al. Elevated blood pressure profile and left ventricular mass in children and young adults with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 3, 1451–1457 (1993).
Bardaji, A. et al. Cardiac involvement in autosomal dominant polycystic kidney disease: a hypertensive heart disease. Clin. Nephrol. 56, 211–220 (2002).
Cadnapaphornchai, M. A. et al. Increased left ventricular mass in children with autosomal dominant polycystic kidney disease and borderline hypertension. Kidney Int. 74, 1192–1196 (2008).
Saggar-Malik, A. et al. Left ventricular mass in normotensive subjects with autosomal dominant polycystic kidney disease. BMJ 309, 1617–1618 (1994).
Bardaji, A. et al. Left ventricular mass and diastolic function in normotensive young adults with autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 32, 970–975 (1998).
Oflaz, H. et al. Biventricular diastolic dysfunction in patients with autosomal dominant polycystic kidney disease. Kidney Int. 68, 2244–2249 (2005).
Verdecchia, P. et al. Circadian blood pressure changes and left ventricular hypertrophy in essential hypertension. Circulation 81, 528–536 (1990).
Li Kam Wa, T. C. et al. Ambulatory blood pressure in hypertensive patients with autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 12, 2075–2080 (1997).
Valero, F. A. et al. Ambulatory blood pressure and left ventricular mass in normotensive patients with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 10, 1020–1026 (1999).
Martinez-Vea, A. et al. Exercise blood pressure, cardiac structure, and diastolic function in young normotensive patients with polycystic kidney disease: a prehypertensive state. Am. J. Kidney Dis. 44, 216–223 (2004).
Almeida, E. A. et al. Tissue Doppler imaging in the evaluation of left ventricular function in young adults with autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 47, 587–592 (2006).
Phillips, R. A. et al. Relation among left ventricular mass, insulin resistance, and blood pressure in nonobese subjects. J. Clin. Endocrinol. Metab. 83, 4284–4288 (1998).
Ohya, Y. et al. Hyperinsulinemia and left ventricular geometry in a work-site population in Japan. Hypertension 27, 729–734 (1996).
Vareesangthip, K. et al. Insulin resistance in adult polycystic kidney disease. Kidney Int. 52, 503–508 (1997).
Lumiaho, A. et al. Insulin resistance is related to left ventricular hypertrophy in patients with polycystic kidney disease type 1. Am. J. Kidney Dis. 41, 1219–1224 (2003).
Rocchini, A. P. et al. Hyperinsulinemia and the aldosterone and pressor responses to angiotensin II. Hypertension 15, 861–866 (1990).
Lembo, G. et al. Abnormal sympathetic overactivity evoked by insulin in the skeletal muscle of patients with essential hypertension. J. Clin. Invest. 90, 24–29 (1992).
Gharavi, A. G. et al. Deletion polymorphism of the angiotensin-converting enzyme gene is independently associated with left ventricular mass and geometric remodeling in systemic hypertension. Am. J. Cardiol. 77, 1315–1319 (1996).
Estacio, R. O. et al. Deletion polymorphism of the angiotensin-converting enzyme gene is associated with an increase in left ventricular mass in men with type 2 diabetes mellitus. Am. J. Hypertens. 12, 637–643 (1999).
Baboolal, K. et al. Association of the angiotensin I converting enzyme gene deletion polymorphism with early onset of ESRF in PKD1 adult polycystic kidney disease. Kidney Int. 52, 607–613 (1997).
Perez-Oller, L. et al. Influence of the ACE gene polymorphism in the progression of renal failure in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 34, 273–278 (1999).
Van Dijk, M. A. et al. The ACE insertion/deletion polymorphism has no influence on progression of renal function loss in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 15, 836–839 (2000).
Schiavello, T. et al. Angiotensin-converting enzyme activity and the ACE Alu polymorphism in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 16, 2323–2327 (2001).
Ecder, T. et al. No effect of angiotensin-converting enzyme gene polymorphism on disease progression and left ventricular hypertrophy in autosomal dominant polycystic kidney disease. Am. J. Nephrol. 23, 466–470 (2003).
Chapman, A. B. et al. Intracranial aneurysms in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 327, 916–920 (1992).
Ruggieri, P. M. et al. Occult intracranial aneurysms in polycystic kidney disease: screening with MR angiography. Radiology 191, 33–39 (1994).
Graf, S. et al. Intracranial aneurysms and dolichoectasia in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 17, 819–823 (2002).
Hadimeri, H. et al. Coronary aneurysms in patients with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 9, 837–841 (1998).
Kanagasundaram, N. S. et al. Aneurysm of the splenic artery in a patient with autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 14, 183–184 (1999).
Torra, R. et al. Abdominal aortic aneurysms and autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 7, 2483–2486 (1996).
Belz, M. M. et al. Familial clustering of ruptured intracranial aneurysms in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 38, 770–776 (2001).
Belz, M. M. et al. Recurrence of intracranial aneurysms in autosomal-dominant polycystic kidney disease. Kidney Int. 63, 1824–1830 (2003).
Schrier, R. W. Optimal care of autosomal dominant polycystic kidney disease patients. Nephrology 11, 124–130 (2006).
Schrier, R. W. et al. Repeat imaging for intracranial aneurysms in patients with autosomal dominant polycystic kidney disease with initially negative studies: a prospective ten-year follow-up. J. Am. Soc. Nephrol. 15, 1023–1028 (2004).
Leier, C. V. et al. Cardiovascular abnormalities associated with adult polycystic kidney disease. Ann. Intern. Med. 100, 683–688 (1984).
Hossack, K. F. et al. Echocardiographic findings in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 319, 907–912 (1988).
Timio, M. et al. The spectrum of cardiovascular abnormalities in autosomal dominant polycystic kidney disease: a 10-year follow-up in a five-generation kindred. Clin. Nephrol. 37, 245–251 (1992).
Lumiaho, A. et al. Mitral valve prolapse and mitral regurgitation are common in patients with polycystic kidney disease type 1. Am. J. Kidney Dis. 38, 1208–1216 (2001).
Klahr, S. et al. Dietary protein restriction, blood pressure control, and the progression of polycystic kidney disease. J. Am. Soc. Nephrol. 5, 2037–2047 (1995).
Maschio, G. et al. Effect of the angiotensin-converting-enzyme inhibitor benazepril on the progression of chronic renal insufficiency. N. Engl. J. Med. 334, 939–945 (1996).
Ecder, T. et al. Effect of antihypertensive therapy on renal function and urinary albumin excretion in hypertensive patients with autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 35, 427–432 (2000).
Chapman, A. B. et al. Overt proteinuria and microalbuminuria in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 5, 1349–1354 (1994).
Van Dijk, M. A. et al. No effect of enalapril on progression in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 18, 2314–2320 (2003).
Zeltner, R. et al. Renal and cardiac effects of antihypertensive treatment with ramipril vs metoprolol in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 23, 573–579 (2008).
Schrier, R. W. et al. Epidemiological study of kidney survival in autosomal dominant polycystic kidney disease. Kidney Int. 63, 678–685 (2003).
Ecder, T. et al. Progress in the blood pressure control in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 36, 266–271 (2000).
Ecder, T. et al. Diuretics versus angiotensin-converting enzyme inhibitors in autosomal dominant polycystic kidney disease. Am. J. Nephrol. 21, 98–103 (2001).
Nutahara, K. et al. Calcium channel blocker versus angiotensin II receptor blocker in autosomal dominant polycystic kidney disease. Nephron Clin. Pract. 99, c18–c23 (2005).
Jafar, T. H. et al. The effect of angiotensin-converting enzyme inhibitors on progression of advanced polycystic kidney disease. Kidney Int. 67, 265–271 (2005).
Ecder, T. et al. Reversal of left ventricular hypertrophy with angiotensin converting enzyme inhibition in hypertensive patients with autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 14, 1113–1116 (1999).
Schrier, R. et al. Cardiac and renal effects of standard versus rigorous blood pressure control in autosomal-dominant polycystic kidney disease: results of a seven-year prospective randomized study. J. Am. Soc. Nephrol. 13, 1733–1739 (2002).
Chapman, A. B. Approaches to testing new treatments in autosomal dominant polycystic kidney disease: insights from the CRISP and HALT-PKD studies. Clin. J. Am. Soc. Nephrol. 3, 1197–1204 (2008).
Schrier, R. W. Renal volume, renin–angiotensin–aldosterone system, hypertension and left ventricular hypertrophy in patients with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. (in press).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R. W. Schrier has acted as a consultant for Amgen and Otsuka and has received grant research/support from Astellas. T. Ecder declared no competing interests.
Rights and permissions
About this article
Cite this article
Ecder, T., Schrier, R. Cardiovascular abnormalities in autosomal-dominant polycystic kidney disease. Nat Rev Nephrol 5, 221–228 (2009). https://doi.org/10.1038/nrneph.2009.13
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2009.13
This article is cited by
-
Autosomal dominant polycystic kidney disease with ectopic unilateral multicystic kidney: a case report
Journal of Medical Case Reports (2024)
-
Efficacy of beetroot juice on reducing blood pressure in hypertensive adults with autosomal dominant polycystic kidney disease (BEET-PKD): study protocol for a double-blind, randomised, placebo-controlled trial
Trials (2023)
-
Cardiovascular implications of hypertensive autosomal dominant polycystic kidney disease: a systematic review and meta-analysis
Pediatric Nephrology (2023)
-
Prevalence of cardiac valvar abnormalities in children and young people with autosomal dominant polycystic kidney disease
Pediatric Nephrology (2023)
-
Generation of heterozygous PKD1 mutant pigs exhibiting early-onset renal cyst formation
Laboratory Investigation (2022)