Abstract
Background
Endogenous opioid peptides have been shown to play a role in the development and/or perpetuation of inflammation. We hypothesize that the endogenous opioid system is involved in inflammatory bowel disease, and antagonism of the opioid–opioid receptor will lead to reversal of inflammation.
Aims
A randomized double-blind placebo-controlled study was designed to test the efficacy and safety of an opioid antagonist for 12 weeks in adults with active Crohn’s disease.
Methods
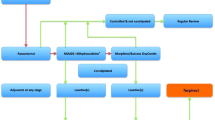
Forty subjects with active Crohn’s disease were enrolled in the study. Randomized patients received daily oral administration of 4.5-mg naltrexone or placebo. Providers and patients were masked to treatment assignment. The primary outcome was the proportion of subjects in each arm with a 70-point decline in Crohn’s Disease Activity Index score (CDAI). The secondary outcome included mucosal healing based upon colonoscopy appearance and histology.
Results
Eighty-eight percent of those treated with naltrexone had at least a 70-point decline in CDAI scores compared to 40% of placebo-treated patients (p = 0.009). After 12 weeks, 78% of subjects treated with naltrexone exhibited an endoscopic response as indicated by a 5-point decline in the Crohn’s disease endoscopy index severity score (CDEIS) from baseline compared to 28% response in placebo-treated controls (p = 0.008), and 33% achieved remission with a CDEIS score <6, whereas only 8% of those on placebo showed the same change. Fatigue was the only side effect reported that was significantly greater in subjects receiving placebo.
Conclusions
Naltrexone improves clinical and inflammatory activity of subjects with moderate to severe Crohn’s disease compared to placebo-treated controls. Strategies to alter the endogenous opioid system provide promise for the treatment of Crohn’s disease.





Similar content being viewed by others
References
Strober W, James SP. The immunopathogenesis of gastrointestinal and hepatobiliary diseases. JAMA. 1992;268:2910–2917.
Fiocchi C. Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology. 1998;115:182–205.
Gurudu S, Fiocchi C, Katz JA. Inflammatory bowel disease. Best Pract Res Clin Gastroenterol. 2002;16:77–90.
Targan SR, Murphy LK. Clarifying the causes of Crohn’s. Nat Med. 1995;1:1241–1243.
Kelly JK, Sutherland LR. The chronological sequence in the pathology of Crohn’s disease. J Clin Gastroenterol. 1988;10:28–33.
Lichtenstein GR, Hanauer SB, Sandborn WJ. Management of Crohn’s disease in adults. Am J Gastroenterol. 2009;104:465–483.
Bewtra M, Lewis JD. Safety profile of IBD: lymphoma risks. Gastroenterol Clin North Am. 2009;38:669–689.
Mackey AC, Green L, Liang LC, Dinndorf P, Avigan M. Hepatosplenic T cell lymphoma associated with infliximab use in young patients treated for inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2007;44:265–267.
Shale M, Kanfer E, Panaccione R, Ghosh S. Hepatosplenic T cell lymphoma in inflammatory bowel disease. Gut. 2008;57:1639–1641.
Toruner M, Loftus EV Jr, Harmsen WS, et al. Risk factors for opportunistic infections in patients with inflammatory bowel disease. Gastroenterology. 2008;134:929–936.
Pol O, Puig MM. Expression of opioid receptors during peripheral inflammation. Curr Top Med Chem. 2004;4:51–61.
Rogers TJ, Peterson PK. Opioid G protein-coupled receptors: signals at the crossroads of inflammation. Trends Immunol. 2003;24:116–121.
Bryant HU, Bernton EW, Holaday JW. Immunosuppressive effects of chronic morphine treatment in mice. Life Sci. 1987;41:1731–1738.
Grimm MC, Ben Baruch A, Taub DD, Howard OM, Wang JM, Oppenheim JJ. Opiate inhibition of chemokine-induced chemotaxis. Ann N Y Acad Sci. 1998;840:9–20.
Janecka A, Fichna J, Janecki T. Opioid receptors and their ligands. Curr Top Med Chem. 2004;4:1–17.
McCarthy L, Wetzel M, Sliker JK, Eisenstein TK, Rogers TJ. Opioids, opioid receptors, and the immune response. Drug Alcohol Depend. 2001;62:111–123.
Kamphuis S, Eriksson F, Kavelaars A, et al. Role of endogenous pro-enkephalin A-derived peptides in human T cell proliferation and monocyte IL-6 production. J Neuroimmunol. 1998;84:53–60.
Vujic V, Stanojevic S, Dimitrijevic M. Methionine-enkephalin stimulates hydrogen peroxide and nitric oxide production in rat peritoneal macrophages: interaction of mu, delta and kappa opioid receptors. Neuroimmunomodulation. 2004;11:392–403.
Peng X, Mosser DM, Adler MW, Rogers TJ, Meissler JJ Jr, Eisenstein TK. Morphine enhances interleukin-12 and the production of other pro-inflammatory cytokines in mouse peritoneal macrophages. J Leukoc Biol. 2000;68:723–728.
Hook S, Camberis M, Prout M, Le Gros G. Absence of preproenkephalin increases the threshold for T cell activation. J Neuroimmunol. 2003;140:61–68.
Zagon IS, McLaughlin PJ. Opioids and differentiation in human cancer cells. Neuropeptides. 2005;39:495–505.
Zagon IS, Jenkins JB, Sassani JW, et al. Naltrexone, an opioid antagonist, facilitates reepithelialization of the cornea in diabetic rat. Diabetes. 2002;51:3055–3062.
Matters GL, Harms JF, McGovern C, et al. The opioid antagonist naltrexone improves murine inflammatory bowel disease. J Immunotoxicol. 2008;5:179–187.
Smith JP, Stock H, Bingaman S, Mauger D, Rogosnitzky M, Zagon IS. Low-dose naltrexone therapy improves active Crohn’s disease. Am J Gastroenterol. 2007;102:820–828.
Best WR, Becktel JM, Singleton JW, Kern F Jr. Development of a Crohn’s disease activity index. National Cooperative Crohn’s Disease Study. Gastroenterology. 1976;70:439–444.
Mary JY, Modigliani R. Development and validation of an endoscopic index of the severity for Crohn’s disease: a prospective multicentre study. Groupe d’Etudes Therapeutiques des Affections Inflammatoires du Tube Digestif (GETAID). Gut. 1989;30:983–989.
Dieleman LA, Palmen MJ, Akol H, et al. Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. Clin Exp Immunol. 1998;114:385–391.
Irvine EJ, Feagan B, Rochon J, et al. Quality of life: a valid and reliable measure of therapeutic efficacy in the treatment of inflammatory bowel disease. Canadian Crohn’s Relapse Prevention Trial study group. Gastroenterology. 1994;106:287–296.
Brazier JE, Harper R, Jones NM, et al. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. BMJ. 1992;305:160–164.
Sandborn WJ, Targan SR. Biologic therapy of inflammatory bowel disease. Gastroenterology. 2002;122:1592–1608.
D’Haens G, van Deventer S, Van Hogezand R, et al. Endoscopic and histological healing with infliximab anti-tumor necrosis factor antibodies in Crohn’s disease: A European multicenter trial. Gastroenterology. 1999;116:1029–1034.
Rutgeerts P, Diamond RH, Bala M, et al. Scheduled maintenance treatment with infliximab is superior to episodic treatment for the healing of mucosal ulceration associated with Crohn’s disease. Gastrointest Endosc. 2006;63:433–442.
Dotan I, Rachmilewitz D, Schreiber S, et al. A randomised placebo-controlled multicentre trial of intravenous semapimod HCl for moderate to severe Crohn’s disease. Gut. 2010;59:760–766.
Schreiber S, Rutgeerts P, Fedorak RN, et al. A randomized, placebo-controlled trial of certolizumab pegol (CDP870) for treatment of Crohn’s disease. Gastroenterology. 2005;129:807–818.
Jones J, Loftus EV Jr, Panaccione R, et al. Relationships between disease activity and serum and fecal biomarkers in patients with Crohn’s disease. Clin Gastroenterol Hepatol. 2008;6:1218–1224.
Froslie KF, Jahnsen J, Moum BA, Vatn MH. Mucosal healing in inflammatory bowel disease: results from a Norwegian population-based cohort. Gastroenterology. 2007;133:412–422.
Devlin SM, Panaccione R. Evolving inflammatory bowel disease treatment paradigms: top-down versus step-up. Gastroenterol Clin North Am. 2009;38:577–594.
Acknowledgments
This research was supported by NIH DK073614 from the National Institutes of Health and IBD-0180R from the Broad Medical Research Program. The University GCRC was funded by M01 RR010732 & C06-RR016499 grants from the National Institutes of Health.
Conflict of interest
Drs. Smith and Zagon have intellectual property rights and have a patent for the use of naltrexone in IBD. This disclosure was provided to all study participants. The statistical analysis of the entire data sets pertaining to efficacy (specifically primary and major secondary efficacy endpoints) and safety (specifically, serious adverse events as defined in federal guidelines) have been independently confirmed by a biostatistician who has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, J.P., Bingaman, S.I., Ruggiero, F. et al. Therapy with the Opioid Antagonist Naltrexone Promotes Mucosal Healing in Active Crohn’s Disease: A Randomized Placebo-Controlled Trial. Dig Dis Sci 56, 2088–2097 (2011). https://doi.org/10.1007/s10620-011-1653-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1653-7